
Beta-Amyloid Plaques (brown) and Tau (blue within neuron) in the Brain In the Alzheimer’s affected brain. Flickr.com, NIH Image Gallery.
Alzheimer's disease and amyloid plaques
Alzheimer's disease is the most common form of dementia. Given its prevalence, it remains the most common cause for neuronal degeneration and death (1).
For many years, a major focus in this disease has been the presence of excess amyloid-beta deposition with the formation of abnormal bundles of protein (neuritic plaques) outside the neurons. It was thought that those plaques were a primary contributor to the development of Alzheimer's dementia (1, 2, 3).
Amyloid plaques are not necessary nor sufficient for the development of Alzheimer's Disease
However, up to one-third of patients with typical Alzheimer's dementia do not have evidence of amyloid plaques (2). And some people who were tested close to the time of death displayed the typical Alzheimer's Dementia pathology in their brain, complete with plaques and excess abnormal Tau protein, but were found to have intact cognitive function (4).
It seems that there are other paths to the development of neuronal death in the brain in Alzheimer's Disease: amyloid plaques do not fit the role of a necessary and sufficient element in the pathophysiology of the disease. Let's look at other intracellular factors that contribute to the pathology of the disease. In this way, it will be easier to appreciate how conditions such as aging, diabetes and obesity can predispose to Alzheimer's Disease, regardless whether amyloid plaques develop or not.
Diabetes and Alzheimer's Disease
Persons with diabetes are unfortunately at much higher risk of having problems with memory, rational judgement ability, and overall cognition (2). The reason is that the brain is highly dependent on glucose as an energy source. It consumes 25% of the glucose in an awake resting state even though its mass is only 2% of the body's total weight.
Insulin and glucose are AWOL in the brain
However, in diabetics the high glucose is not available for use by neurons for several reasons:
- There are lower levels of insulin in the brain. Of note, those lower levels also contribute to an increase in appetite, compounding insulin resistance
- There are less glucose transporters in the neuronal bodies and synaptic membranes (GLUT3 and GLUT4 proteins, respectively)
- There are lower levels of glucose in the cerebrospinal fluid (dysfunction of the blood-brain-barrier)
- There are lower levels of insulin receptors
- There are abnormalities in the lipids in the membranes (leading to defective insulin receptor signaling)
- Astrocytes are not able to produce glycogen properly (and the number of astrocytes in diabetic brains are reduced)
In summary, the brains of people with diabetics are characterized by:
- A decreased availability of insulin and
- Lower levels of glucose available to the cells. The brain is forced to use alternate sources of energy
Lower levels of insulin in an Alzheimer's Disease brain.....
An aging brain, a diabetic brain, and a brain affected by Alzheimer's disease are all sorely deficient in insulin. Not only are the insulin receptors reduced, but the insulin concentration in the cerebrospinal fluid is reduced (2).
Less insulin and less insulin receptors are a problem. Insulin triggers neuronal neurite outgrowth, synaptic density, and neuronal survival. Insulin is a neurotrophic factor for the brain.
...and the hippocampus goes awry
In fact, insulin has been shown to promote learning and memory. This is because insulin is directly related to optimal hippocampus function and memory potentiation (2, 5, 6). Experimentally, administration of insulin has been shown to improve performance in memory tasks. Conversely, blocking insulin function in the hippocampus directly leads to cognitive deficits (6). Finally, intranasal insulin seems to enhance memory in healthy patients as well as those afflicted with Alzheimer's Disease (5).
The brain needs insulin signaling through its receptors to maintain its normal function (2, 5, 6). In particular, the hippocampus - the site of learning and memory - is critically dependent on glucose and insulin (6). This is why many researchers describe Alzheimer's disease as "Type 3 diabetes”, where affected patients have the same biochemical abnormalities in their brains as diabetics have in their pancreas and muscles.
Lower glucose levels, a hypometabolic brain, .....
Interestingly, in Alzheimer's Disease, there is evidence of hypometabolic changes in the brain 10 years before the appearance of dementia symptoms.
Other conditions of hypometabolism in the brain also predispose to cognitive deficits. Thus, thiamine deficiency (Beriberi/chronic alcoholism), Down's Syndrome, patients who carry the ApoE4 gene, Parkinson's disease, acquired epilepsy, and diabetics all show evidence of hypometabolic states in their brains (2, 7, 8, 9).
...and higher use of alternative energy sources
Deficit of glucose (and insulin) means that the brain will inexorably go down a path of dysfunctional energy production:
- The brain will resort to other molecules to produce energy, such as breaking down myelin to produce fatty acids and ketones, which are shuttled into producing Acetyl CoA (to produce energy through oxidative phosphorylation)
- Astrocytes can't produce glycogen in conditions of relative insulin deficiency. This compounds the dysfunctional energy systems
- Moreover, the neurons are asking for more lactate to use as fuel, since they don't have glucose; lactate is shuttled into producing more Acetyl CoA (to produce energy through oxidative phosphorylation)
- Astrocytes also have decreased access to glucose; this means that they cannot process glutamate. Increased glutamate levels can be toxic to cells
To compound the injury, several of the enzymes that produce energy in a brain affected by Alzheimer's disease are dysfunctional. This means that the production of ATP is inefficient. This leads to increased oxidative damage to the cell as the cells can't maintain healthy metabolic systems. A vicious cycle ensues of abnormal and oxidized proteins leading to decreased efficiency in the production of ATP, which then leads to the generation of increasing numbers of disordered proteins (3, 9, 10).
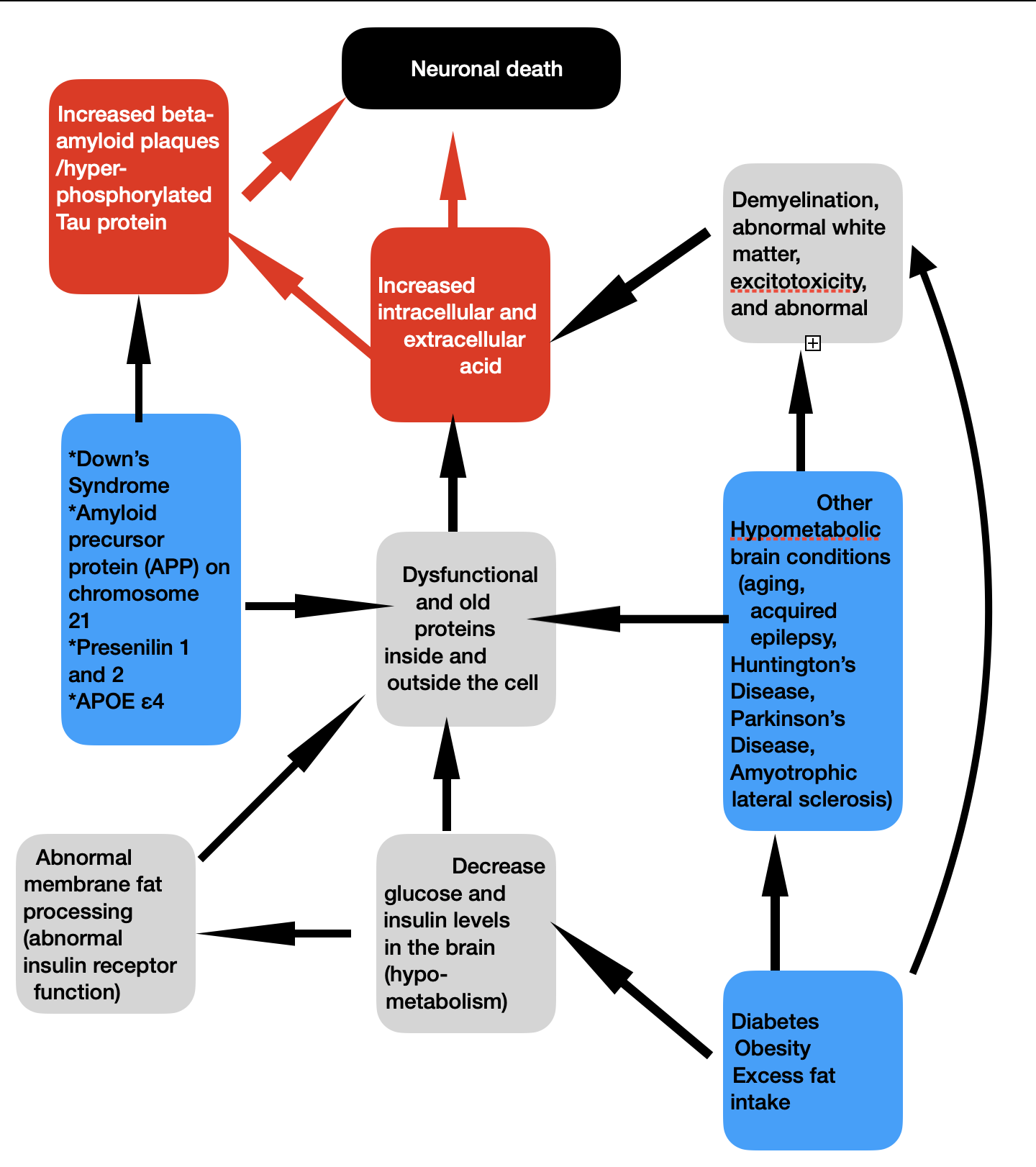
Hijab J. Alzheimer's Disease and pathophysiology of neuronal death. April 2023
Alzheimer's Disease: Running the engine on empty
Why does using alternate sources of fuel cause problems for neuronal cells? You'd think the cells can figure out low glucose levels and plan for it.
There are several reasons. Keep in mind that it is not just 1 or 2 weeks that the cells have to deal with running on empty: it is decades. Over this span of time:
- The cells are degrading myelin to use the fat for extra energy molecules
- The cells are continuously revved up, trying to produce ATP with dysfunctional oxidative enzymes
- Lactate is being shuttled to the Oxidative cycle. This means it is not available to help shuttle H+ out of the neuron
- Astroglia are overproducing glutamic acid, which becomes excitotoxic
- Astroglia can't take a break. This means that they are spending all their energy on providing for the neuron, rather than taking a breather and proliferating
Acidifying the brain
Unfortunately, all those processes lead to dysfunctional acid processing inside and outside the neuronal cell. Interestingly, generating ATP is one of the mechanisms that cells have for consuming H+. Thus, defective ATP production also contributes to the acidosis (11).
Studies have shown that there is a pathological lowering of the pH in neurons in many neurodegenerative diseases (13). Not only that, the extracellular space is also abnormally acidic in Alzheimer's disease, Amyotrophic lateral sclerosis, and Huntington's disease ((12).
Such dysregulated H+ processing leads to defective neuronal transmission, leading to many dysfunctional processes, including lower acetylcholine levels at the synapses. Medications such as acetylcholinesterase inhibitors do not address the underlying pathology of Alzheimer's dementia. The problem is much more widespread, involving critical pathways that help keep a healthy homeostasis within the neuron's cell body, its axons, the myelin sheath, and the supporting astroglia.
Back to Amyloid plaques
Studies have shown that accumulation of beta-amyloid is induced by intracellular acidosis. Conversely, increasing the pH within the cytoplasm (alkalization) will cause the beta-amyloid plaques to stop aggregating (2,12).
Clearly processes that induce changes in the metabolic profile of the neuronal cytoplasm will be sufficient to jumpstart amyloid plaque deposition outside the neuron. As shown in the figure above, there are multiple roads that leads to the distinctive pathology for which Alzheimer's disease is renowned. But they all seem to start from a defective cytoplasmic engine within neurons (2, 3, 8, 9, 10, 12, 13).
References
- Erkkinen MG, Kim MO, Geschwind MD. Clinical Neurology and Epidemiology of the Major Neurodegenerative Diseases. Cold Spring Harb Perspect Biol. 2018 Apr 2;10(4):a033118. doi: 10.1101/cshperspect.a033118. PMID: 28716886; PMCID: PMC5880171.
- Yoon JH, Hwang J, Son SU, Choi J, You SW, Park H, Cha SY, Maeng S. How Can Insulin Resistance Cause Alzheimer's Disease? Int J Mol Sci. 2023 Feb 9;24(4):3506. doi: 10.3390/ijms24043506. PMID: 36834911; PMCID: PMC9966425.
- Butterfield DA, Halliwell B. Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci. 2019 Mar;20(3):148-160. doi: 10.1038/s41583-019-0132-6. PMID: 30737462; PMCID: PMC9382875.
- Iacono D, Resnick SM, O'Brien R, Zonderman AB, An Y, Pletnikova O, Rudow G, Crain B, Troncoso JC. Mild cognitive impairment and asymptomatic Alzheimer disease subjects: equivalent β-amyloid and tau loads with divergent cognitive outcomes. J Neuropathol Exp Neurol. 2014 Apr;73(4):295-304. doi: 10.1097/NEN.0000000000000052. PMID: 24607960; PMCID: PMC4062187.
- Dou JT, Chen M, Dufour F, Alkon DL, Zhao WQ. Insulin receptor signaling in long-term memory consolidation following spatial learning. Learn Mem. 2005 Nov-Dec;12(6):646-55. doi: 10.1101/lm.88005. Epub 2005 Nov 14. PMID: 16287721; PMCID: PMC1356184.
- McNay EC, Ong CT, McCrimmon RJ, Cresswell J, Bogan JS, Sherwin RS. Hippocampal memory processes are modulated by insulin and high-fat-induced insulin resistance. Neurobiol Learn Mem. 2010 May;93(4):546-53. doi: 10.1016/j.nlm.2010.02.002. Epub 2010 Feb 20. PMID: 20176121; PMCID: PMC2878207.
- Zammit MD, Laymon CM, Tudorascu DL, Hartley SL, Piro-Gambetti B, Johnson SC, Stone CK, Mathis CA, Zaman SH, Klunk WE, Handen BL, Cohen AD, Christian BT. Patterns of glucose hypometabolism in Down syndrome resemble sporadic Alzheimer's disease except for the putamen. Alzheimers Dement (Amst). 2021 Jan 13;12(1):e12138. doi: 10.1002/dad2.12138. PMID: 33490360; PMCID: PMC7804861.
- Zilberter Y, Zilberter M. The vicious circle of hypometabolism in neurodegenerative diseases: Ways and mechanisms of metabolic correction. J Neurosci Res. 2017 Nov;95(11):2217-2235. doi: 10.1002/jnr.24064. Epub 2017 May 2. PMID: 28463438.
- Blázquez E, Hurtado-Carneiro V, LeBaut-Ayuso Y, Velázquez E, García-García L, Gómez-Oliver F, Ruiz-Albusac JM, Ávila J, Pozo MÁ. Significance of Brain Glucose Hypometabolism, Altered Insulin Signal Transduction, and Insulin Resistance in Several Neurological Diseases. Front Endocrinol (Lausanne). 2022 May 9;13:873301. doi: 10.3389/fendo.2022.873301. PMID: 35615716; PMCID: PMC9125423.
- Demetrius LA, Simon DK.Biogerontology. An inverse-Warburg effect and the origin of Alzheimer's disease. 2012 Dec;13(6):583-94. doi: 10.1007/s10522-012-9403-6. Epub 2012 Oct 20.PMID: 23086530
- Silverstein TP. The Proton in Biochemistry: Impacts on Bioenergetics, Biophysical Chemistry, and Bioorganic Chemistry. Front Mol Biosci. 2021 Nov 26;8:764099. doi: 10.3389/fmolb.2021.764099. PMID: 34901158; PMCID: PMC8661011.
- Harguindey S, Alfarouk K, Polo Orozco J, Reshkin SJ, Devesa J. Hydrogen Ion Dynamics as the Fundamental Link between Neurodegenerative Diseases and Cancer: Its Application to the Therapeutics of Neurodegenerative Diseases with Special Emphasis on Multiple Sclerosis. Int J Mol Sci. 2022 Feb 23;23(5):2454. doi: 10.3390/ijms23052454. PMID: 35269597; PMCID: PMC8910484.
- Schwartz L, Henry M, Alfarouk KO, Reshkin SJ, Radman M. Metabolic Shifts as the Hallmark of Most Common Diseases: The Quest for the Underlying Unity. Int J Mol Sci. 2021 Apr 12;22(8):3972. doi: 10.3390/ijms22083972. PMID: 33921428; PMCID: PMC8068795.
